Publications
There is a continually updated listing of all publications on both Pubmed and Google Scholar. Request reprints of any of these articles: contact Dr. Sacks.
Wembo Zhang | Giovanni Rossini | David Kamensky | Tan Bui-Thanh | Michael S. Sacks
The functional complexity of native and replacement aortic heart valves (AVs) is well known, incorporating such physical phenomenons as time‐varying non‐linear anisotropic soft tissue mechanical behavior, geometric non‐linearity, complex multi‐surface time varying contact, and fluid–structure interactions to name a few. It is thus clear that computational simulations are critical in understanding AV function and for the rational basis for design of their replacements. However, such approaches continued to be limited by ad‐hoc approaches for incorporating tissue fibrous structure, high‐fidelity material models, and valve geometry. To this end, we developed an integrated tri‐leaflet valve pipeline built upon an isogeometric analysis framework. A high‐order structural tensor (HOST)‐based method was developed for efficient storage and mapping the two‐dimensional fiber structural data onto the valvular 3D geometry. We then developed a neural network (NN) material model that learned the responses of a detailed meso‐structural model for exogenously cross‐linked planar soft tissues. The NN material model not only reproduced the full anisotropic mechanical responses but also demonstrated a considerable efficiency improvement, as it was trained over a range of realizable fibrous structures. Results of parametric simulations were then performed, as well as population‐based bicuspid AV fiber structure, that demonstrated the efficiency and robustness of the present approach. In summary, the present approach that integrates HOST and NN material model provides an efficient computational analysis framework with increased physical and functional realism for the simulation of native and replacement tri‐leaflet heart valves.
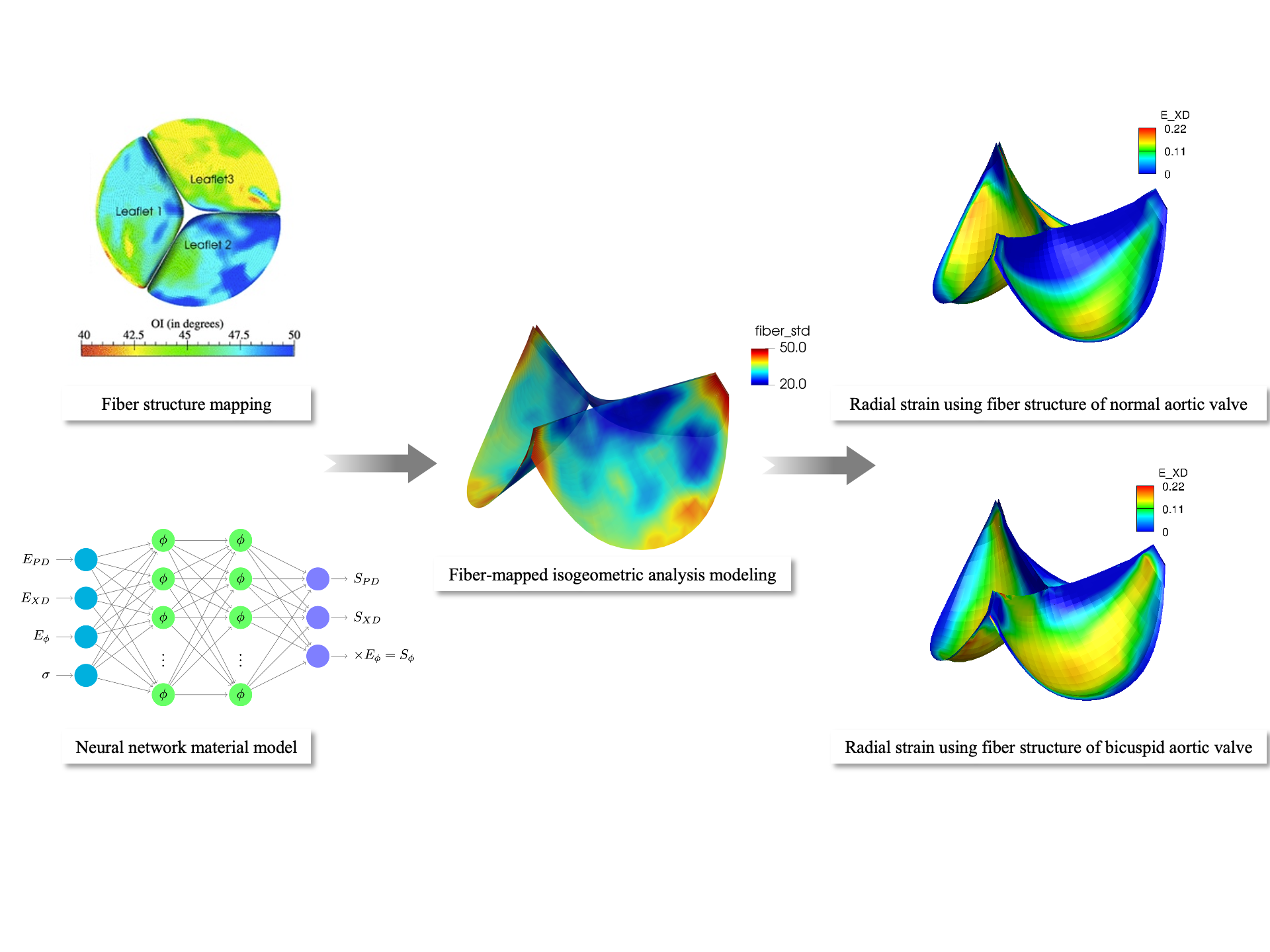
“Isogeometric finite element‐based simulation of the aortic heart valve: Integration of neural network structural material model and structural tensor fiber architecture representations - Zhang - 2021 - International Journal for Numerical Methods in Biomedical Engineering - Wiley Online Library.” https://onlinelibrary.wiley.com/doi/full/10.1002/cnm.3438
Harshita Narang | Bruno V. Rego | Amir H. Khanlighi | Ahmed Aly | Alison M. Pouch | Robert C. Gorman | Joseph H. Gorman III | Michael S. Sacks
Ischemic mitral regurgitation (IMR) is a prevalent cardiac disease associated with substantial morbidity and mortality. Contemporary surgical treatments continue to have limited long-term success, in part due to the complex and multi-factorial nature of IMR. There is thus a need to better understand IMR etiology to guide optimal patient specific treatments. Herein, we applied our finite element-based shape-matching technique to non-invasively estimate peak systolic leaflet strains in human mitral valves (MVs) from in-vivo 3D echocardiographic images taken immediately prior to and post-annuloplasty repair. From a total of 21 MVs, we found statistically significant differences in pre-surgical MV size, shape, and deformation patterns between the with and without IMR recurrence patient groups at 6 months post-surgery. Recurrent MVs had significantly less compressive circumferential strains in the anterior commissure region compared to the recurrent MVs (p = 0.0223) and were significantly larger. A logistic regression analysis revealed that average pre-surgical circumferential leaflet strain in the Carpentier A1 region independently predicted 6-month recurrence of IMR (optimal cutoff value - 18%, p = 0.0362). Collectively, these results suggest greater disease progression in the recurrent group and underscore the highly patient-specific nature of IMR. Importantly, the ability to identify such factors pre-surgically could be used to guide optimal treatment methods to reduce post-surgical IMR recurrence.
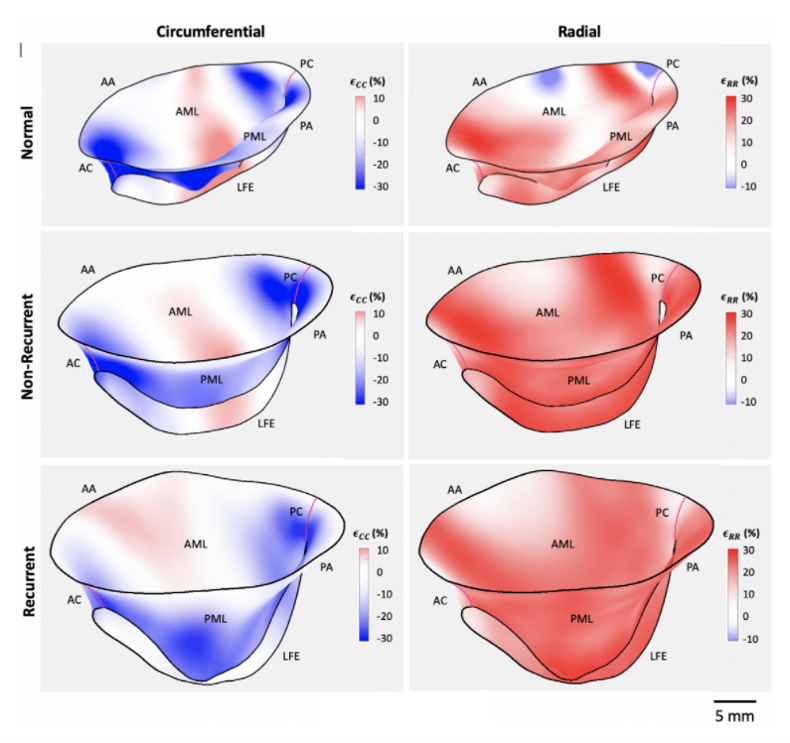
H. Narang et al., “Pre-surgical Prediction of Ischemic Mitral Regurgitation Recurrence Using In Vivo Mitral Valve Leaflet Strains,” Ann. Biomed. Eng., Apr. 2021, doi: 10.1007/s10439-021-02772-5. https://link.springer.com/article/10.1007/s10439-021-02772-5